Abstract
Background Literature supports the need for improved techniques to achieve spinopelvic alignment and reduce complication rates in patients with adult spinal deformity (ASD). Personalized interbody devices were developed to address this need and are under evaluation in the multicenter Clinical Outcome Measures in Personalized aprevo Spine Surgery (COMPASS) registry. This report presents interim COMPASS pre- and postoperative sagittal alignment results and complication rates for a subcohort of COMPASS patients diagnosed and surgically treated for spinal deformity.
Methods COMPASS is a postmarket observational registry of patients enrolled either before or after index surgery and then followed prospectively for 24 months. Sagittal alignment was assessed with SRS-Schwab modifiers for pelvic incidence minus lumbar lordosis, pelvic tilt, and T1 pelvic angle. Summed SRS-Schwab modifiers were utilized to assign overall deformity status as mild, moderate, or severe. Complications were extracted from patient medical records.
Results The study included 67 patients from 9 centers. Preoperative severe deformity was observed in 66% of patients. Index surgeries included implantation of a median of 2 personalized interbody devices by anterior, lateral, or transforaminal approaches and with a median of 8 posteriorly instrumented levels. Overall postoperative sagittal alignment improved with a significant decrease in the mean sum of SRS-Schwab modifiers that correlated strongly to improvements in pelvic incidence minus lumbar lordosis. Among 44 patients with preoperative severe overall deformity, 16 improved to moderate and 9 to mild deformity. Complications occurred for 13 patients (19.4%), including 1 mechanical complication requiring revision 9 months after surgery and none related to personalized interbody devices.
Conclusions This study demonstrates that ASD patients whose treatment included personalized interbody devices can obtain favorable postoperative alignment status comparable to published results and with no complications related to the personalized interbody devices.
Clinical Relevance This study contributes to growing evidence that personalized interbody devices contribute to improved sagittal alignment in ASD patients by directly adjusting the orientation of adjacent vertebra.
Level of Evidence 3.
Introduction
Historically, patients treated surgically for spinal deformity have had high rates of new or persistent postoperative malalignment and complications requiring revision surgery. To address these issues, personalized interbody devices have been developed to predictably control the disc space and the resulting segmental and regional spinal alignment. As with any new medical technology, it is important to study the effectiveness and outcomes. Toward this objective, the multicenter Clinical Outcome Measures in Personalized aprevo Spine Surgery (COMPASS) observational registry has been established. This ongoing registry enables the collection of baseline, operative, and follow-up data on patients treated with spinal fusion that included personalized interbody devices. The overall objective of this registry effort is to assess whether the utilization of personalized interbody devices in the surgical treatment of spinal deformity and/or degenerative pathologies will improve the ability to achieve alignment goals, reduce the risk of interbody subsidence, enhance fusion rates through improved implant-to-endplate contact, and reduce revision rates due to mechanical complications. This report presents interim COMPASS results for a subcohort of COMPASS patients diagnosed and surgically treated for spinal deformity.
While surgical treatment has been shown to be superior to conservative care for adult spinal deformity (ASD) patients,1 the incidence of adverse events (AEs) and long-term complications is high: Up to 75% of patients still have a radiographic deformity after surgery,2 over 40% experience a major AE that is surgery or implant related,3 and over 20% require revision surgery.4 For example, Glassman et al showed that among 122 operative ASD patients, 38 (31%) had revision surgery by year 5 postoperatively, 3 patients had 2 revisions, and 1 patient had 3 revisions.5 The costs of complications include emotional and physical distress for patients and their families along with the significant financial burden to the healthcare system.6
AEs that result in revision surgery are primarily mechanical complications including proximal junctional kyphosis (PJK), pseudoarthrosis, cage subsidence, and rod breakage. PJK has been associated with overcorrection and lordotic disproportion.7 Pseudarthrosis can result from suboptimal loading conditions, specifically the load distribution and contact area between the vertebral endplates and the interbody bone graft. Endplate subsidence can lead to loss of correction as well as pseudoarthrosis. Rod failure can be the result of extreme rod bends or excessive loading over time. When the intervertebral bone fails to fuse, anatomical loads are transferred to the spinal rod, exposing it to excessive loading over time and an increased likelihood of dynamic rod fracture.8
Although the focus of this report is on deformity patients, spinal alignment is equally important for surgical patients with degenerative pathologies. Over the past decade, there has been increasing evidence linking the presence of postoperative spinal malalignment in these patients to higher risk for adjacent segment disease requiring surgery.7,9,10 Residual postoperative sagittal malalignment in degenerative patients is also a risk factor for mechanical failures, including PJK, that require revision surgery.11 Collectively, current literature strongly supports the need for improved approaches and techniques to better achieve spinopelvic alignment following spine fusion.
This report presents interim results from the COMPASS observational registry for a cohort of patients who underwent surgical treatments to address ASD. The purpose of this study is to compare pre- and postoperative radiographic sagittal spinopelvic alignment parameters and track complications in ASD patients whose treatment included personalized interbody devices.
Methods
Creation of Personalized Interbody Devices
Personalized interbody devices (aprevo, Carlsmed, Carlsbad, CA) are cleared by the US Food and Drug Administration, are individually designed for each patient, and are 3D-printed from titanium alloy (Ti-6Al-4V). Three-dimensional spine models are rendered from patient computed tomography and radiographic images and used to specify device dimensions and contours that match and adjust the orientation of adjacent vertebral endplates. The ordering surgeon reviews and approves specifications for planned intervertebral anterior and posterior height along with intervertebral lordotic and coronal angles for each fused level. Personalized devices are available for anterior, lateral, and transforaminal surgical approaches (Figure 1).
Personalized lumbar interbody devices for anterior lumbar interbody fusion, lateral lumbar interbody fusion, and transforaminal lumbar interbody fusion.
Study Design
COMPASS is a postmarket observational registry of patients surgically treated with personalized lumbar interbody devices. Participating patients are enrolled before or after their index surgery and then followed prospectively at intervals of 6 weeks, 6 months, 12 months, and 24 months postoperatively. All 16 participating US centers obtained Institutional Review Board approval prior to study initiation, and all patients provided informed consent.
The present study includes COMPASS patients for whom surgical treatment addressed spinal deformity defined radiographically by the presence of 1 or more moderate or severe sagittal modifiers or coronal deformities according to the SRS/Schwab ASD classification.12 Thresholds for moderate or severe deformity include sagittal measurements of pelvic incidence minus lumbar lordosis (PI−LL) ≥10°, pelvic tilt (PT) ≥20°, or T1 pelvic angle (T1PA) ≥10° and coronal angles greater than 30° for thoracic, thoracolumbar, or double curves.12–14 Eligible patients underwent index surgeries including 1 or more personalized interbody devices with at least 3 instrumented fusion levels and had pre- and postoperative radiographs available for measurement of spinopelvic alignment parameters.
Radiographic Assessment
The most recent anteroposterior and lateral full-length radiographs of each patient were provided to a central site for radiographic measurements using validated software (SpineView, ENSAM Laboratory of Biomechanics, Paris, France).15
Spinopelvic parameters, including PI−LL, PT, and T1PA, were used to assess deformity severity utilizing the modifiers from the SRS-Schwab classification for ASD (Table 1).12,13 The T1PA modifier, a validated surrogate for sagittal vertical axis (SVA) in the SRS-Schwab classification, represents global sagittal alignment independent of pelvic compensatory mechanisms and thus is a reflection of both PT and SVA.14 T1PA is also readily available from most full-length lateral spine radiographs, including uncalibrated images. The sum of the SRS-Schwab sagittal alignment modifiers was then used to categorize the deformity severity as mild, moderate, or severe for each patient (Table 2), as previously described.16,17
Definitions for SRS-Schwab modifiers and severity thresholds for sagittal deformity.
Definition of the sum of SRS-Schwab modifiers.
AEs and Complications
AEs were assessed for severity based on National Institutes of Health guidelines for serious adverse event (SAE) definitions from the National Institute of Arthritis and Musculoskeletal and Skin Diseases.18 Complications for this study were defined as SAEs that include any death, life-threatening event, an event that caused significant or permanent disability, or an event that resulted in prolonged or new hospitalization.19 Occurrences, treatments, and outcomes for AEs were extracted from patient medical records. AEs and complications were categorized as medical or surgical according to the International Spine Study Group-AO Spine complication classification system.20,21 Complications were further categorized by (a) occurrence either ≤90 days postoperatively, including intraoperatively, or >90 days postoperatively and (b) for relatedness to a personalized interbody device. Mechanical complications were identified as SAEs resulting from adjacent segment disease including proximal/distal junctional kyphosis and/or implant failure or dislodgement that resulted in reoperation.17,20,22,23
Statistical Methods
Statistical analysis was performed using SPSS software version 29.0.2.0. Demographic, radiographic, and operative data were assessed with descriptive statistics using mean and SD or median and range for continuous data, depending on the distribution and frequencies with percentages for categorical variables. Pre- and postoperative data were compared using a paired t test for continuous variables and nonparametric Wilcoxon signed-rank test for ordinal data. Correlations of each SRS-Schwab modifier with the SRS-Schwab modifier sum were performed using Spearman’s rank correlation. All tests were 2-tailed with a P value <0.05 considered significant.
Results
Cohort Overview
Study data collection for 67 eligible patients occurred at 9 centers (5 academic and 4 nonacademic). Descriptive data for these COMPASS deformity patients are summarized in Table 3. The majority (70%) of patients were women. The mean age was 66.2 ± 10.1 years, and the mean body mass index was 28.6 ± 5.7 kg/m2. Half (50%) of patients had a history of previous spine surgery. The mean preoperative spinopelvic alignment measurements exceeded thresholds for moderate or severe deformity for all 3 SRS-Schwab modifiers (Table 3).
Demographics, spine surgery history, and preoperative radiographic parameters of the study population.
The median postoperative follow-up from surgery was 14.7 months and ranged from 3.9 to 39.2 months (Table 4). Forty patients had more than 12-month follow-up, and 9 patients exceeded 24-month follow-up. Surgeries included 4 to 18 posteriorly instrumented vertebra. Among the patients, 43.3% underwent posterior-only approaches with transforaminal lumbar interbody fusion devices, whereas 56.7% had anterior lumbar interbody fusion, lateral lumbar interbody fusion, or combined approaches with anterior lumbar interbody fusion and lateral lumbar interbody fusion devices (Table 4). Interbody fusions included personalized interbody devices at L5 to S1 in 42 cases and L4 to S1 in 26 cases. Eleven patients received 1 or more additional nonpersonalized interbody devices.
Operative parameters of the study population.
Radiographic Alignment
Spinopelvic alignment parameters were measured from preoperative and most recent postoperative sagittal radiographs for each patient. Postoperative radiographic measurements were available from follow-up patient visits at 6 weeks (n = 11), 6 months (n = 30), 12 months (n = 24), and 24 months (n = 2).
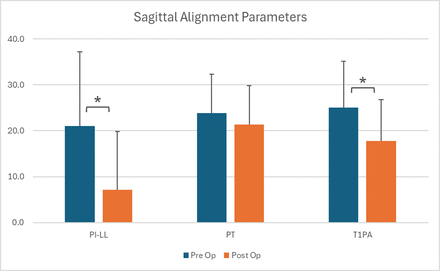
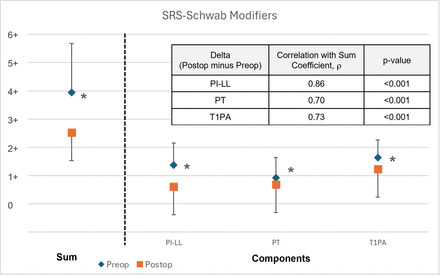
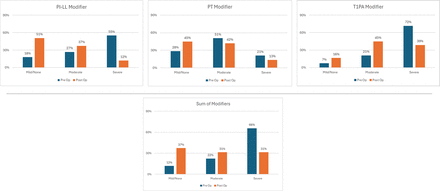
Pre- and postoperative alignment measurements are summarized in Figure 2. Mean preoperative PI−LL decreased significantly from 21.0° ± 16.2° to 7.1° ± 12.7° postoperatively (P < 0.001). Similarly, T1PA decreased from 25.0° ± 10.1° to 17.7° ± 9.0° (P < 0.001), whereas PT did not change significantly. These trends are reflected in the decreases in mean SRS-Schwab modifiers for PI−LL and T1PA, as well as the significant drop in the mean sum of SRS-Schwab modifiers (Figure 3). The sum of SRS-Schwab modifiers correlated strongly to the PI−LL modifier and moderately to PT and T1PA modifiers (Figure 3). Patient distributions of SRS-Schwab component modifiers are summarized in Figure 4. The percentage of patients starting in the severe (++) PI−LL modifier category was reduced from 55% preoperatively to 12% postoperatively. While only 12 preoperative patients (18%) were categorized as mild (0), 34 patients (51%) met this criterion postoperatively. Similarly, preoperative severe T1PA modifier distribution decreased from 72% to 39% of patients, and T1PA mild modifiers increased from 7% to 16%. This is consistent with the distribution of the summed modifier scores that decreased from 66% to 31% of patients with severe (4+ to 6+) and increased from 12% to 37% of patients with mild (0 to 1+) summed scores.
Radiographic sagittal alignment measurements for 67 study patients. Mean postoperative pelvic incidence minus lumbar lordosis (PI−LL) and T1 pelvic angle (T1PA) were significantly reduced compared with preoperative measurements.
Pre- vs postoperative sum of SRS-Schwab modifiers and component SRS-modifiers (mean ± SD). The postoperative change (delta) in pelvic incidence minus lumbar lordosis (PI−LL) modifier has the strongest correlation to the change in the sum of modifiers.
Pre- vs postoperative patient distributions of SRS-Schwab component modifiers and sum of modifiers. Data presented are the percentage of patients.
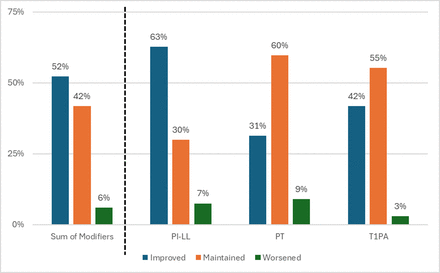
Directional changes in patient deformity status are represented in Figure 5, with a change in status represented by a modifier change from one score to another. PI−LL modifiers were improved in 63% of patients and maintained in 30%. PT modifiers were improved in 31% of patients and maintained in 60%. T1PA modifiers were improved in 42% and maintained in 55%. Directional changes to summed modifiers in this study were improved for 52% of patients, maintained for 32%, and worsened for 6%.
Distribution of patients according to directional postoperative changes in sagittal SRS-Schwab alignment modifier scores for pelvic incidence minus lumbar lordosis (PI−LL), pelvic tilt (PT), and T1 pelvic angle (T1PA). Data presented are the percentage of patients with improved, maintained, or worsened postoperative modifier scores.
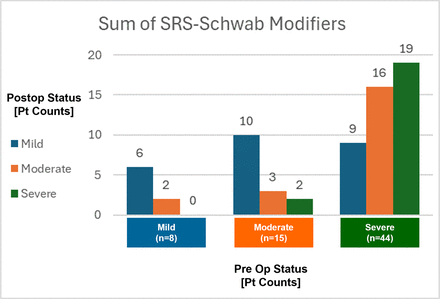
Both magnitude and directional changes in patient deformity status are represented in Figure 6. Among 44 patients with preoperative severe sum of modifiers scores, 9 improved to mild, 16 improved to moderate, and 19 maintained severe. Preoperative moderate scores improved to mild for 10 of 15 patients, and preoperative mild scores were maintained in 6 of 8 patients. The sum of modifier scores worsened for 4 of 67 patients in the study cohort.
Postoperative changes in magnitude and direction of summed modifiers for 67 study patients.
Eight patients (12%) had preoperative mild/no deformity according to the SRS-Schwab modifier classification, as shown in the lower panel of Figure 4 and the preoperative mild status group in Figure 6. These patients were diagnosed and treated for coronal plane malalignment. Sagittal correction was not a treatment priority for these patients, and the resulting postoperative sum of modifier scores was either maintained (n = 6) or slightly worsened to moderate (n = 2).
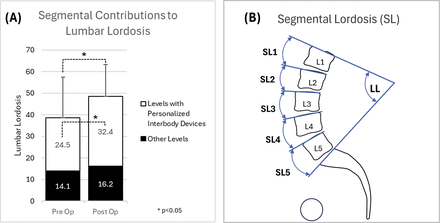
The reduction in mean postoperative PI−LL was primarily due to the 10° increase in mean postoperative LL (P < 0.05). Lumbar lordosis is the sum of segmental lordosis angles between L1 and S1. The contribution to LL from levels treated with personalized interbody devices is represented in Figure 7 and compared with the contribution from other levels. The postoperative mean sum of segmental angles increased by 7.9° (P < 0.05) for levels treated with personalized devices.
Contribution of segmental corrections to changes in lumbar lordosis for 67 study patients (A). Lumbar lordosis is the sum of segmental lordosis angles between L1 and S1 (B). “Other Levels” include (a) levels with posterior instrumentation and fusion only, (b) levels treated with nonpersonalized interbody devices, and (c) levels not treated.
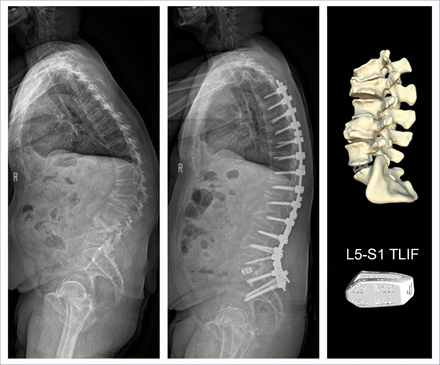
Two patient examples with pre- and postoperative imaging along with personalized interbody device designs are shown in Figures 8 and 9.
Baseline and 6-month radiographs of a 77-year-old man who underwent L2–S1 posterolateral fusion with lateral lumbar interbody fusion (LLIF) interbody devices at L3–L5.
Baseline and 6-month radiographs of a 59-year-old man who underwent L5–S1 transforaminal lumbar interbody fusion (TLIF) and posterolateral fusion from T4 to the pelvis.
Complications
AE reports and complications are listed in Table 5 and include nonserious AEs and complications. Complications occurred for 13 patients (19.4%), including 2 medical and 10 surgical complications ≤90 days postoperatively and 1 surgical complication >90 days postoperatively. Two patients (3%) with surgical complications required reoperation ≤90 days postoperatively to address screw malposition or loosening. One mechanical complication with PJK required reoperation 9 months postoperatively. No complications directly related to the personalized interbody devices were identified.
AEs and complication SAEs.
Discussion
Although surgery for ASD can offer significant clinical improvement, these procedures have high rates of complications and risk of revision surgery. Postoperative persistent or new spinal malalignment is a significant risk factor for revision surgery, and improved techniques are needed to facilitate better postoperative alignment. The primary tools to correct spinal deformity are osteotomies and interbody releases and fusions. Personalized interbody devices offer the potential to specifically control alignment of the disc space, which may result in more predictable achievement of segmental, regional, and goal spinal alignment. The COMPASS registry prospectively follows patients treated with personalized interbody devices to assess their alignment and outcomes. In the present study, we provide an interim assessment of ASD patients enrolled in COMPASS to date. We demonstrate that the alignment achieved with personalized interbody devices compares favorably with previous reports, and no complications in the series were directly attributable to the personalized devices.
PI−LL stands out as the alignment parameter most affected by surgical intervention in this study population as demonstrated by the significant decrease of 13.9° in mean postoperative PI−LL and improved PI−LL modifiers in 63% of patients, resulting in improved summed modifiers in 52% of patients. This outcome is attributable to the increase of 7.9° in mean postoperative lumbar lordosis, resulting from segmental corrections at levels treated with personalized interbody devices. These results are corroborated by data from Sadrameli et al who describe the achievement of planned corrections to intervertebral lordosis in 82% of lumbar fusion levels treated with personalized interbody devices.24 Anterior column realignment is typically a priority during ASD surgery, and data from this COMPASS series demonstrate that personalized interbody devices may directly and controllably adjust lumbar lordosis.
Among the 67 patients in this study, 26 (38%) improved from preoperative moderate (+) or severe (++) PI−LL modifiers to PI−LL modifier zero. This compares to results by Moal et al who reported that 13% of patients improved from preoperative moderate or severe PI−LL modifiers to modifier zero.2 The majority of lumbar lordosis occurs in the distal lumbar spine (L4–S1), and this study included 42 cases (63%) with personalized interbody devices implanted at L4 to L5 or L4 to S1. T1PA also improved postoperatively, although to a lesser extent than PI−LL mismatch. This may be due to the modest changes to PT in study patients, and T1PA is related in part to PT and SVA as a measure of global alignment.
Directional changes to component SRS-Schwab modifiers in the current study are comparable to published results by Passias et al who reported that PI−LL modifiers were improved in 46% of patients and maintained in 47%, PT modifiers were improved in 28% and maintained in 59%, and SVA modifiers were improved in 53% and maintained in 39%.22
This study included 8 patients (12%) who presented with primary coronal plane malalignment. COMPASS surgeon investigators chose to address deformity in these patients with personalized interbody devices designed to match anatomic contours and adjust the orientation of adjacent vertebral endplates in their plans to surgically improve or maintain alignment. A worsened postoperative sum of SRS-Schwab modifiers was observed for 4 subjects (6%), which could be due to the discrete thresholds for SRS-Schwab modifiers such that small changes in continuous angular measurements result in ordinal shifts to a higher modifier state. For comparison, Passias reported between 7% and 12% of patients had worsened postoperative SRS-Schwab component modifiers.22
Complications in the study include a reoperation rate of 3% during the 90-day postoperative period that can be cautiously compared with the 7% reoperation rate for the 6-week postoperative period recently published for a larger ASD cohort of 1260 patients.20 There were low rates of reoperation in the current study, including 1 revision due to a mechanical complication that occurred 9 months postoperatively. Recent publications report rates of reoperation for mechanical complications between 10% and 32% over postoperative periods of 1 to 2 years.17,20,23 Importantly, there were no complications in this study attributable to the personalized interbody devices. These findings are favorable, albeit preliminary, and will be updated in subsequent reports on a larger cohort with longer follow-up.
Although the present study benefits from the prospective multicenter design and the inclusion of both academic and nonacademic surgeon practices, there are limitations. Since this is an interim assessment of an ongoing registry study, the length of postoperative follow-up is limited and varies among participating subjects, although the majority of patients have achieved at least 1-year follow-up. In addition, the number of patients limits the granularity of assessments that can be performed at the present time. The chosen SRS-Schwab classification for this study is appropriate for sagittal alignment assessment but does not assess radiographic outcomes for patients with primarily coronal deformities. Future studies of COMPASS patients should include rigorous analysis of coronal deformity parameters.
Conclusion
This study demonstrates that ASD patients whose surgical treatment included personalized interbody devices can obtain postoperative alignment status during the early postoperative period that is at least comparable to published results without complications directly attributable to the personalized interbody devices. As patient enrollment continues to increase and follow-up matures, we anticipate the ability to more definitively assess the potential short- and long-term benefits of personalized interbody devices.
Footnotes
Funding This work was supported by funding from Carlsmed.
Declaration of Conflicting Interests Roland S. Kent discloses that he is a clinical research investigator and receives consulting fees from Carlsmed. Christopher P. Ames discloses that he is a clinical research investigator and receives consulting fees from Carlsmed. Jahangir Asghar discloses that he is a clinical research investigator and receives consulting fees from Carlsmed. Donald J. Blaskiewicz discloses that he is a clinical research investigator and receives consulting fees from Carlsmed. Joseph A. Osorio discloses that he is a clinical research investigator and receives consulting fees from Carlsmed. Chun-Po Yen discloses that he is a clinical research investigator and receives consulting fees from Carlsmed. Jeffrey P. Mullin discloses that he is a clinical research investigator and receives consulting fees from Carlsmed. Justin S. Smith discloses that he is a shareholder and receives consulting fees from Carlsmed. John M. Small discloses that he is a clinical research investigator for Carlsmed. Michele Temple-Wong discloses that she is an employee of Carlsmed. Jeffrey D. Schwardt discloses that he is an employee of Carlsmed.
Disclosures In addition to the relationships listed in the Declaration of Conflicting Interests, Christopher Ames reports grants/contracts from SRS; royalties/licenses from DePuy Synthes, K2M, Next Orthosurgical, Stryker, Biomet Zimmer Spine, Medicrea, and NuVasive; consulting fees from DePuy Synthes, Medicrea, Agada Medical, Medtronic, and K2M; is the chair of the SRS Safety and Value Committee and serves on the executive committee of ISSG; serves on the editorial board fro Operative Neurosurgery and Neurospine; is the director of Global Spinal Analytics, and has research interests with Titan Spine, ISSG, and DePuy Synthes. Roland Kent reports consulting fees from SI Bone and Globus Medical; payment/honoraria from SI Bone, Globus Medical, and Premia Spine; and patents pending for SI Bone. Jeffrey Mullin reports consulting fees from Medtronic, Globus, and SI Bone, and serves on the CNS and AANS/CNS Joint Spine Section Executive Committees. Joseph Osorio reports grants/contracts from Medtronic; royalties/licenses from Alphatec; and consulting fees from Alphatec, Medtronic, and DePuy. John Small reports royalties/licenses from Astura Spine. Justin Smith reports grants/contracts from SeaSpine/Orthofix, NREF, AO Spine, and DePuy Synthes/ISSGF; royalties/licenses from Highridge and Globus/NuVasive; consulting fees from Highridge, SeaSpine/Orthofix, Medtronic, Cerapedics, and Globus/NuVasive; support for attending meetings/travel from AO Spine; serving on the SRS Board of Directors and ISSGF Executive Committee; and stock/stock options from Alphatec and Globus/NuVasive. Chun-Po Yen reports consulting fees from Life Spine and Medtronic and support for teaching a cadaveric course from Life Spine.
Ethics Statement Institutional Review Board approval was obtained prior to patient enrollment. All patients provided informed consent.
- This manuscript is generously published free of charge by ISASS, the International Society for the Advancement of Spine Surgery. Copyright © 2024 ISASS. To see more or order reprints or permissions, see http://ijssurgery.com.