Abstract
Lateral lumbar interbody fusion (LLIF) is an advantageous approach for spinal arthrodesis for a wide range of spinal disorders including degenerative, genetic, and traumatic conditions. LLIF techniques have evolved over the past 15 years regarding surgical approach, with concomitant improvements in implant material design. Bioactive materials have been a focus in the development of novel methods, which reduce the risk of subsidence and pseudarthrosis. Historically, polyetheretherketone and titanium cages have been selected for their advantageous biomechanical properties; however, both have their limitations, regarding optimal modulus or osseointegrative properties. Recent modifications to these 2 materials have focused on devising bioactive implants, which may enhance the rate of bony fusion in spinal arthrodesis by addressing the shortcomings of each. Specific emphasis has been placed on developing improvements in surface coating, porosity, microroughness, and nanotopography of interbody cages. This has been coupled with advances in additive manufacturing to generate cages with ideal biomechanical properties. Three-dimensional-printed titanium cages may be particularly beneficial in spinal arthrodesis during LLIF and reduce the historical rates of subsidence and pseudarthrosis by combining a number of these putatively beneficial biomaterial properties.
INTRODUCTION
Lateral lumbar interbody fusion (LLIF) is an excellent option for the treatment of degenerative spinal conditions as it allows for restoration of the normative alignment, lordosis, and height of diseased spinal segments.1 The anterolateral surgical corridors enable placement of large interbody grafts while simultaneously allowing for annulus release and thorough disc space preparation. The ability to place large grafts that span the apophyseal ring enables anterior column load bearing sufficient to restore disc space collapse, decrease the risk of subsidence, and achieve indirect decompression of the neural elements.2 The ability to resist cage settling and obtain high fusion rates has been demonstrated in repeated studies.3–5
Over the past 15 years since the first report of the lateral interbody fusion, there has been a tremendous amount of literature published supporting its use. Simultaneously, there have been great advances in interbody technology, in both the cervical and lumbar spine. Recent developments have been targeted at improving integration of the implant with the bone-implant interface to allow for better fusion rates and enhanced biomechanical stability. The expansion of material sciences and additive manufacturing technology has further augmented the advances in the development of new products, birthing the concept of “bioactive” implants. The goals of these bioactive implants are to allow for improved initial structural support through appositional ongrowth/ingrowth and also to play a stimulatory and facilitative role in bone healing and fusion. In this review, we examine the evolution of these implants in the context of LLIF surgery.
IMPLANT CAGES
Polyetheretherketone
Early adoption of the LLIF was performed with the use of polyetheretherketone (PEEK) cages. Based on extensive data from the cervical and lumbar interbody literature, PEEK has the advantage of containing a modulus of elasticity similar to that of cortical bone (Table).6 Although originally developed in the 1980s for industrial applications, specifically aeronautical use in the blades of turbines and aircraft, the use of PEEK was not applied to spinal fusion until the 1990s. By using a purely synthetic material with properties similar to that of bone, surgeons realized they could obviate the need for obtaining structural autograft, thereby decreasing risks related to additional procedures or increased blood loss. Further, the radiolucency of the semicrystalline linear polycyclic aromatic thermoplastic permits radiographic assessment of fusion progress. With broad application for transforaminal and anterior lumbar interbody fusion, it was a natural first material to be coupled with the introduction of the miniopen LLIF procedure.1
Comparison of implant characteristics among different materials utilized in interbody cages.
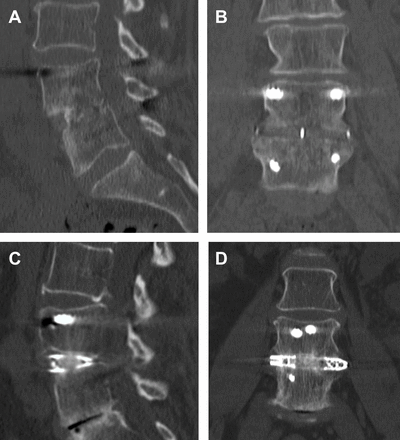
Early designs of PEEK interbody grafts included large central channels in which bone graft could be placed, with the intention of bony growth through these channels to connect each vertebra directly. Given the radiolucent nature of the grafts, interbody arthrodesis with these implants could easily be assessed on postoperative radiographs by evaluating the presence of bone contained within channels spanning from rostral to caudal end plates (see Figure 1a and b). Combined fusion rates from multiple studies with PEEK interbodies for transpsoas LLIF have been reported to be approximately 93%, with radiographic subsidence noted in approximately 14% of levels.12 Risk factors for subsidence have been suggested to include surgical variables such as end plate injury and the inclusion of supplemental fixation, patient factors such as bone density, and also specific implant characteristics such as cage width.13–15 Studies with PEEK cages have demonstrated that greater anterior-posterior width resulted in a decreased incidence of subsidence.16 This highlights the role of these interbody spacers as mechanical struts that overcome settling by distributing loads over their large surface area.
The 1-y sagittal (A, C) and coronal (B, D) computed tomography postoperative images demonstrating lumbar lateral interbody fusion with polyetheretherketone (A, B) (Coroent XL, Nuvasive, San Diego, CA) and 3-dimensional-printed titanium cages (C, D) (Modulus, Nuvasive, San Diego, CA).
Successful interbody fusion requires bone growth between the vertebral bodies, which is achieved through the primary bony remodeling phenomenon of osteogenesis, osteoinduction, and osteoconduction. However, PEEK is a biologically inert substance without the ability to significantly stimulate any of these processes, and in fact, can be inhibitory. As such, bone growth can only occur through (via the channels in the implant) or around the implant. The implant’s main purpose is to act as a biomechanical spacer that maintains the space between the end plates while contained allograft within the channels bridges the bony end plates. It is well documented that haloing around the PEEK implants may occur.17 Fibrous capsule formation around the implant initiates the cascade, in turn resulting in further micromotion, inflammation, and osteolysis.18 Should substantial bone growth through or around the implant not occur, implant failure and pseudarthrosis will inevitably result. This shortcoming has opened the door for newly designed bioactive implants that aim to provide added stabilization at the graft-end plate interface through the process of osseointegration.19
Titanium
Titanium presents itself as an exciting alternative to PEEK implants for several reasons. On a macroscopic level, it has a favorable weight-to-strength ratio and supports use in interbody implants resisting large compressive loads. Just as beneficial, on a biological level, titanium implants form TiO2 which enhances bony ongrowth of the graft surface. This is believed to occur by generating hydroxide ions, which promote adsorption of Ca2− and PO4 3− molecules, in turn forming apatite and stimulating osteoblasts.20,21 Essentially, this oxide layer mimics the chemical ceramic properties of hydroxyapatite (HA) and results in fortuitous osteoblastic activity.
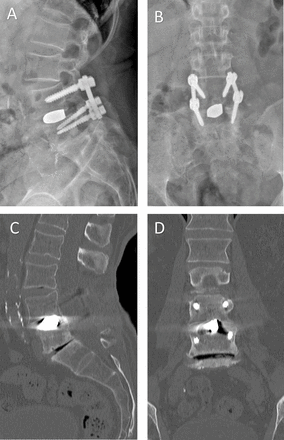
Unfortunately, titanium has a high modulus of elasticity relative to bone. The modulus of elasticity for cortical bone is roughly 18 GPa. Pure PEEK has a modulus of approximately 4 GPa, but with the addition of carbon fiber to its mixture, can be closely matched to that of cortical bone.7 Titanium is significantly greater (110 GPa) and therefore represents an increased risk of stress shielding and resultant interbody subsidence (Table).22 Systematic reviews of titanium vs PEEK implants for posterior interbody fusion have demonstrated that titanium cages resulted in higher reported fusion rates, but with increased risk of subsidence.23 However, these studies utilized block titanium structures, and with the advent of architectural adjustments in titanium devices that have a structural stiffness that can be modified, the material modulus of elasticity alone is likely not reflective of the implant behavior in vivo. Three-dimensional (3D) titanium structures and the effective modulus or stiffness can thus be formulated to match more closely that of bone. A potential downside to titanium cages is difficulty in interpreting postoperative imaging modalities. For example, magnetic resonance imaging and associated artifact can challenge assessment of tissues adjacent to the implants, and titanium’s radiopacity makes plain radiographs less desirable for evaluation of bony fusion (Figure 2).
Lateral (A) and anteroposterior (B) x-ray imaging and sagittal (C) and coronal (D) computed tomography images of a patient treated with a block titanium transforaminal lumbar interbody implant 4 y prior. Note the subsidence of the superior end plate of L5 and the extensive radiographic artifact making evaluation of arthrodesis difficult. The absence of haloing around the pedicle screws to suggest screw loosening makes fusion seem probable, and dynamic imaging from this patient showed no excessive motion.
IMPLANT MODIFICATIONS
In addition to selection of an appropriate interbody material, recent studies have evaluated the potential for surface coating to improve surface adhesion/apposition and improve arthrodesis. The native properties of PEEK and titanium, as described above, are favorable for their biomechanical properties; however, they do not inherently promote osseointegration. A significant amount of our understanding in this area comes from the dental and oral maxillofacial implants field. Coatings such as HA or titanium maintain the benefits of the ideal cage elastic modulus while synergistically providing an improved surface for bony fusion via chemical integration. Studies have investigated the use of HA as a surface coating to improve cancellous and cortical bony fusion during spine surgery. HA was first introduced in animal models utilizing titanium-coated pedicle screws by Hasegawa et al.24 Additional animal studies have since demonstrated improved bone-implant interface and strength with HA-coated screws compared to titanium screws alone.25
Surface coating techniques include electron beam melting of materials to improve osseointegrative properties.26 Given the ideal modulus of PEEK, it was posited to combine a PEEK cage with titanium coating to improve bony fusion following spinal arthrodesis. Walsh et al demonstrated plasma-sprayed titanium coating of PEEK cage reduced its hydrophobic nature and promoted bone ongrowth at the bone-implant interface.27 This in turn resulted in dramatically improved shear strength at the bone-implant interface at 4 weeks in their ovine model, suggesting that direct bone to implant bonding may improve mechanical properties of the fusion as well. Titanium surface bonding, however, is not without its downsides as implant impaction during interbody insertion may not withstand shear loading. Kienle et al demonstrated that titanium-coated PEEK cages were prone to loss of some, if not all, of the titanium coating following impaction. In comparison, surface-etched titanium cages were resistant to any surface damage.28
Surface coating of interbodies is not the only modifiable factor that affects the development of interbody fusion. Additional characteristics such as porosity, microroughness,29 and nanotopography19 may be optimized to augment successful bony fusion. Surface topography has been demonstrated to be a key role in the surface chemistry, which permits improved osseointegration of interbody implants. Torstick et al demonstrated that porosity, independent of the graft material, afforded improved osseointegration compared to smooth implants and plasma-sprayed titanium.30,31 Altogether, these individual material properties need to be more extensively studied in the setting of spinal arthrodesis, but should continue to be an important consideration for future development of novel cage designs.
Finally, recent advances related to additive manufacturing processes have allowed for the careful crafting of titanium implants that have altered structural biomechanical properties. Specifically, 3D printing applies computer design to titanium to create implants with an internal architecture that results in a modulus similar to bone. This cleverly utilizes a structurally sound lattice work that provides sufficient strength to withstand loading combined with a modulus that reduces the risk of subsidence, thereby addressing the major shortcoming of traditional solid titanium cages. Additionally, these cages were printed with the intention to provide the porosity necessary to encourage appositional ongrowth/ingrowth or integration, as well as bony fusion. McGilvray et al utilized an in vivo ovine model of lumbar fusion to compare the biomechanical and biologic properties of PEEK, plasma-sprayed porous titanium-coated PEEK, and 3D-printed porous titanium alloy cages. Micro-computed tomography (CT) evaluation demonstrated increased bony fusion mass within the graft of 3D-printed porous titanium cages compared to that of plasma spray-coated PEEK implants. Furthermore, 3D-printed grafts had increased resistance to range of motion on flexion and extension, suggesting the bony ingrowth provided a more clinically effective fusion compared to that of PEEK or plasma-sprayed porous titanium-coated PEEK.32
BIOACTIVE IMPLANTS FOR LATERAL INTERBODY FUSION
For decades prior to the minimally invasive lateral interbody fusions using PEEK implants, open retroperitoneal anterolateral fusions performed typically utilized locally collected autograft tissue. However, it is important to note that some early reports dating back 2 decades suggested using Bagby and Kuslich (BAK)-threaded titanium cages and demonstrated successful fusion (>90%) for this purpose.33 Moreover, in 2007, Takemoto et al used a canine retroperitoneal anterolateral fusion model to demonstrate the value of porous titanium implants over untreated nonporous titanium to the same end.34 Nevertheless, it wasn’t until many years later that titanium implants were again attempted for miniopen lateral fusions.
Multiple recent clinical studies have looked at the efficacy of bioactive implants for modern lateral interbody fusion, specifically the application of 3D-printed titanium grafts. Given the limitations of pure PEEK and titanium implants, there has been a significant interest in designing an interbody cage that combines the modulus nearer to that of cortical bone while providing an osseointegrative surface to optimize bony fusion. Combining these biomechanical and bioactive factors may ultimately reduce the risk of subsidence, which could otherwise result in loss of indirect decompression, loss of lordosis, pseudarthrosis, and potential need for reoperation (Figure 1c and d). Krafft et al reviewed a cohort of patients who underwent LLIF and compared the rates of subsidence in postoperative radiographs. Initial data demonstrated that patients with 3D-printed titanium cages had a decreased rate of subsidence compared to PEEK implants alone, suggesting 3D-printed titanium cages may be a superior implant in LLIF.35 Similarly, Van Horn et al used an ovine model to evaluate and compare the osseointegrative properties of 3D-printed titanium, titanium alloy, and PEEK cages. These sheep studies demonstrated significantly more bony ongrowth with the 3D-printed titanium cages compared to PEEK and titanium alloy cages.36 Further evidence to support the synergy between implant composition and topography in bony fusion is seen in a recent study by Fogel et al, which utilized titanium cages with microporous end plates and internal lattice structure. Using an ovine model of lumbar lateral interbody fusion, these microporous titanium cages with lattice demonstrated a reduction in cage stiffness with segmental stability at 12 weeks following interbody fusion surgery.37
More precise studies assessing the use of stand-alone LLIF with 3D-printed titanium cages found these to be more resistant to severe subsidence (50%–100% loss of postoperative disc height)38 when compared to PEEK alone.39 Specifically, a retrospective review by Adl Amini et al evaluated patients who underwent TLIF with either titanium or PEEK cage and found a significantly decreased rate of early subsidence (6–12 months postsurgery) with 3D-printed titanium cages compared to PEEK cages.39 These findings are complemented by studies which demonstrate PEEK may be directly related to subsidence. Specifically, Satake et al demonstrated an association between PEEK and late settling,15 as well as a correlation between PEEK and intraoperative end plate injury,40 suggesting an increased risk of nonunion. These studies are further supported by a retrospective review of 113 patients, which also found a higher incidence of subsidence with PEEK interbody cages compared to that of titanium alone.8 While the evidence supporting the use of 3D-printed titanium cages is promising, it should be noted that there may be increased associated manufacturing costs. As additive manufacturing techniques improve and become more widely available over time, it is likely that these costs will decrease. Future studies focused on cost-effectiveness may help to delineate whether the added value that decreased subsidence levels brings outweighs these increased upfront implant costs.
As we continue to move forward with bioactive devices, one of the priorities will be to identify optimal means for assessing surface osteointegration and fusion with 3D-printed titanium cages. While motion evaluation of fusion has been utilized on dynamic x-ray imaging in the past, thin-cut CT remains the gold standard for monitoring interbody fusion. Given that bone-implant surface growth provides biomechanical support, traditional assessment of fusion by solely examining bony ingrowth through the channels of a PEEK cage on plain radiographs is not reliable and may suggest artificially low rates of arthrodesis. A recent algorithm for evaluating fusion with these cages has been proposed using CT imaging.9 In this classification, growth and apposition to the implant surface are evaluated in addition to growth through and around the cage to capture the true rate of fusion. Using this grading scheme for assessment of arthrodesis will help to standardize clinical outcomes assessment as the field progresses and more data become available comparing outcomes for novel bioactive implants.
CONCLUSION
Taken together, the current literature demonstrates significant advancements in the material sciences which have provided considerable improvement in our currently available implant materials. A greater appreciation for the value of osseointegration between the implant and the vertebral end plates is developing as mounting evidence supports its importance in spinal fusion. Surface chemistry, roughness, and porosity can be combined with modern additive manufacturing to create implants that have optimal mechanical and biological properties. Recent literature from lateral interbody fusion application of these implants further supports its clinical value. Future large-scale comparative studies are required to demonstrate the impact on clinical and radiological outcomes between various implants.
Footnotes
Funding The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclosures Julie L. Chan reports research materials and funding from Misonix, Inc. Hyun W. Bae reports grants/research support from SpineArt, NuVasive, Medtronic, DePuy, Diffusion Technologies, LDRSpine, Prosidyan, Stryker, and Zimmer; ownership interest/shareholder for Surgery Center; and royalty/patent holder for Biomet, DePuy, LDRSpine, NuVasive, Prosidyan, Stryker, and Zimmer. S. Harrison Farber has no disclosures. Juan S. Uribe reports receiving consulting fees and royalties from Nuvasive and consulting fees from SI Bone and Misonix. Robert K. Eastlack reports receiving royalties from Globus Medical, NuVasive, Seaspine, Aesculap, and SI Bone; being on the speakers bureau for Radius; being a paid consultant company/supplier for NuVasive, Aesculap, K2M-Stryker, SI Bone, Seaspine, Medtronic, Carevature, Spinal Elements, ControlRad, and Biedermann-Motech; receiving stock ownership or stock options (company/supplier) from Alphatec Spine, NuVasive, Seaspine, and Spine Innovations; receiving research support (company/supplier) as a primariy investigator from NuVasive, Seaspine, Medtronic/Titan, and Scripps Clinic Medical Group; other financial or material support (company/supplier) from NuVasive, Seaspine, and AONA; and board member/committee appointments for the Society Of Lateral Access Surgery (SOLAS), Scoliosis Research Society (SRS), San Diego Orthopaedic Society (SDOS)—President, San Diego Spine Foundation—Board member/CFO.
- This manuscript is generously published free of charge by ISASS, the International Society for the Advancement of Spine Surgery. Copyright © 2022 ISASS. To see more or order reprints or permissions, see http://ijssurgery.com.