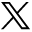Abstract
Background While first-generation articulated disc prostheses had an ideal positioning schematically as posterior as possible because of their geometrically determined center of rotation, the dogma may change for viscoelastic implants, whose center of rotation is free. Our hypothesis was to assess whether the anteroposterior positioning (APP) of a viscoelastic implant may influence the clinical or radiological outcomes at follow-up.
Methods Twenty-five patients (mean age 47 years) were evaluated, with an average follow-up of 25.9 months. The primary outcome was the implants’ APP on lateral radiographs. APP between 0% and 49% meant anterior centering, 50% perfect centering, and 51% to 100% posterior centering. The cohort was divided into 2 groups: anterior positioning and posterior positioning. Measurements were performed blindly to the functional outcomes. Visual analog scale for neck pain and radicular pain and the Neck Disability Index were assessed. Range of motion was measured at the last follow-up. The C2 to C7 Cobb angle and the spinocranial angle were also measured.
Results The median crude offset from the vertebral endplate center was 0.4 mm (mean: 0.3 mm, Q1: −1.5 mm, Q3: 2 mm; range, −2.9 to 4 mm). The mean overall APP was 49%, 45.2% (95% CI, 43.2%–47.1%) in the anterior group, and 54.1% (95% CI, 51.4%–55.3%) in the posterior group. Fifteen patients were in the group anterior positioning and 10 in the group posterior positioning. The mean spinocranial angle was 79° preoperatively and 74° preoperatively (P = 0.04). Functional outcomes were significantly improved at the last follow-up (P < 10−4). There was no significant correlation between the APP, functional outcomes, and range of motion.
Conclusion The APP of the CP-ESP viscoelastic disc arthroplasty does not significantly influence the clinical or radiological outcomes at follow-up. This study suggests that this type of implant tolerates greater variability in its implantation technique.
Level of Evidence 4.
- cervical disc arthroplasties
- unconstrained prostheses
- anteroposterior positioning
- functional outcomes
- sagittal alignment
Introduction
Cervical disc arthroplasties (CDAs) are more and more prevalent and represent an alternative to anterior cervical decompression and fusion for the treatment of radiculopathy and myelopathy.1,2 The principle of anterior cervical decompression and fusion is blocking a functional spinal unit (FSU), and this technique exposes patients to a risk of adjacent disc degeneration.3 Conversely, CDA aims to preserve FSU mobility, and exposes difficulties while setting the center of rotation and restoring alignment. The challenge is even more complex because the instantaneous center of rotation depends on the level4 and varies due to age-related degeneration.5,6 Furthermore, there is a wide variability of type of sagittal alignment for the neck,7 and kyphosis represents one-third of the asymptomatic population. The motion of the cervical spine involves axial rotation, flexion-extension, translation, and lateral bending, and the actual motion of the spine is coupled.8 Consequently, tracking the precise axes of rotation is an exceedingly challenging task.9
Mechanical interlocking prostheses constitute the first generation of such devices, with various types according to numerous parameters, including the geometry of the interlocking or the number of pieces involved in the construct, varying from 1 to 3.10,11 Different designs provide different postimplantation kinematics related to their degree of constraints.12 Unconstrained devices hope to eliminate the difficulty of standardizing the center of rotations and providing better postoperative kinematics;13,14 however, they expose patients to more instability and anterior extrusion of the polyethylene nucleus.15–18 More constrained mechanisms are more stable but may fail to restore the motion19 by fixing the center of rotation.
The second generation of prostheses differs by the nature of the bearing inserted, which is viscoelastic and deformable instead of using a mechanical bearing. This type remains constrained by the elastic return,11,20 but the center of rotation is self-adaptive, and shocks are absorbed and dissipated.11,21 Early evaluations suggest that viscoelastic bearing provides better restoration of the FSU elasticity.11
In current practice, specifically with using the first generation of mechanical interlocking implants, cervical disc replacement is a delicate and demanding surgery that requires careful preparation of the endplates to set its position.22 The uncus processes are reliable anatomical landmarks that help the surgeon center the device laterally.23 However, the setting of the depth is more complicated to achieve. It depends on the quality of the exposition and the release.22,23 Therefore, the viscoelastic devices may be helpful, being permissive and authorizing a more comfortable range of positioning. There are still very limited data supporting this hypothesis. In particular, there is no information about the correlation between the depth of the implant, its instantaneous translation, and patients’ clinical outcomes.
The CP-ESP disc prosthesis (Spine Innovations, France) is a one-piece cervical prosthesis with a viscoelastic core, designed to provide good postoperative kinematics, cervical FSU elasticity, and shock absorption.24 This study aimed to assess the extent to which the depth of the CP-ESP disc prosthesis was associated with patients’ clinical outcomes in a retrospective single-center cohort. The hypothesis was that there was no significant association.
Materials and Methods
Study Design and Setting
This study assesses the outcome of a monocentric retrospective cohort of patients who sustained a single-level CDA with the CP-ESP device. The surgeries were performed by several spine surgeons in a teaching hospital. The period of recruitment began in January 2017 and finished in June 2021. The index date was the surgery date for the follow-up calculation. This study is reported in line with the STROCSS criteria.
Participants
The patients were older than 18 years and sustained a CDA for radiculopathy or myelopathy. All the surgeries were performed through a left anterolateral cervicectomy. Caspar pins distract the interbody space for exposure and disc preparation. The position was checked using trials before the definitive implantation of a CP-ESP prosthesis. This is a one-piece implant with a viscoelastic polycarbonate urethane central core, robustly fixed to 2 T-40 titanium endplates by one male and one female peg. The external part of the endplates is covered by a layer of hydroxyapatite and contains anchoring pegs to improve osteointegration. The implant is available with 3 thicknesses and 3 anteroposterior dimensions. The design of this implant is based on the same principle of the “silent block bush” developed for the lumbar LP-ESP prostheses,25 meaning that there is no center of rotation controlled by the implant itself. The viscoelastic core and this silent block bush allow 6 degrees of freedom (axial rotation, flexion-extension, translation, and lateral bending) and the restoration of the elastic resistance of the FSU.11,20,24
Postoperatively, patients had a soft collar to ease the immediate postoperative pain and were authorized to move without restriction when the pain was controlled.
Variables
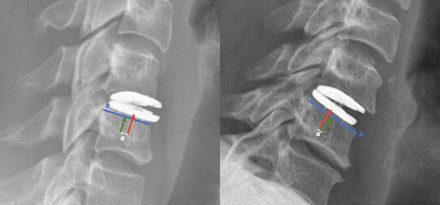
The primary outcome was the measurement of the anteroposterior positioning (APP) of implants on lateral radiographs. The crude offset distance between the center of the inferior endplate of the implant and the center of the upper plateau of the inferior vertebra was measured in millimeters (Figure 1). A positive value meant a posterior offset, a negative value indicated an anterior offset, and 0 conveyed no offset. Then, to obtain an indicator independent of the radiographic scale and inter-individual variations, the distance was relative to the total length of the upper plateau of the inferior vertebra. The APP was thus a ratio, expressed as a percentage; 0% to 49% meant anterior centering, 51% to 100% posterior centering, and 50% perfect centering (reference). The measures were made on the lateral postoperative radiographs in a neutral position using the Picture Archiving and Communications System software (Eastman Kodak/Carestream Health, Rochester, NY, USA). The patients were retrospectively divided into 2 groups according to their APP: the anterior and posterior groups.
Measurement of anteroposterior positioning. The distance between the center of the prosthesis (red arrow) and the center of the endplate of the inferior vertebra (green arrow reference), relative to the total length of the inferior endplate (distance b, blue line). When the ratio was <50%, the prosthesis was positioned anteriorly (image on the left). When the ratio was >50%, the prosthesis was positioned posteriorly (image on the right).
The functional assessment was made using different scales. The visual analog scale (VAS) for neck pain (VAS-N) and radicular pain (VAS-R) were recorded preoperatively and at the last follow-up (LFU) date. These 2 scores range from 0 (no pain) to 10 (excruciating pain). The Neck Disability Index (NDI),26 validated in France,27 was also assessed preoperatively and at LFU. This score contains 10 questions, each ranked from 0 to 5, so the score is ranged from 0 (no disability) to 50 (maximal disability). The improvement of functional outcomes after surgery was assessed by computing the difference between the LFU and preoperative score for NDI, VAS-N, and VAS-R, expressed as a percentage.
Range of motion (ROM) was measured postoperatively after 6 weeks. The sagittal alignment in the neutral position was assessed locally by the mean of the Cobb angle of the FSU, the C2 to C7 Cobb angle, and the spinocranial angle (SCA).7 Per Le Huec et al, the SCA is supposed to be a constant with an average of 83° ± 9°.
The instantaneous translation of the superior endplate on the inferior endplate was measured in the neutral position. It was the distance between the center on the superior endplate and the center of the inferior endplate relative to the total length of the lower endplate and expressed as a percentage. A positive value meant anterior translation of the upper endplate; a negative value meant posterior translation.
The development of heterotopic ossification (HO) was assessed at LFU on lateral radiographs using McAffee’s classification28 in 5 stages, from 0 (no HO) to IV (bony ankylosis).
Data Source Bias
The data were collected retrospectively according to the medical records. The measurements were performed blindly to the functional outcomes. The missing values related to the study’s retrospective nature were a source of bias. To reduce the number of missing values and improve robustness, every patient was phoned to double-check the functional outcome history and perform a new NDI assessment. A multiple imputations using Multivariate Imputation by Chained Equation approach was performed to reconstruct the missing values. A total of 10 datasets were produced, and the analysis was performed on the “native” dataset with the missing values, for complete case analysis, and on the 10 datasets with imputation. A sensitivity analysis was conducted for every model to assess the effect of the unobserved data. Outcomes were considered robust if coefficients and P values remained similar under the 10 alternative scenarios.
The second source of bias identified was the quality of the radiographs, particularly partially rotated lateral radiographs, resulting in the wrong APP assessment. To control this risk, we considered the erect radiographs when the patient could move and not the immediate postoperative radiographs. We verified the quality of the radiographs and the absence of a double-edge aspect of the prosthesis. In case of doubt, the measure was repeated with other radiographs of further follow-up.
Statistical Methods
When describing the database, we reported means and SDs for the quantitative variables and the counts for the categorical variables. Differences in quantitative outcomes between the preoperative period and the LFU (functional outcomes and neutral sagittal alignment parameters) were analyzed using paired Student t tests.
The main hypothesis was that there was no significant association between the APP and the functional outcomes. The associations were assessed using 2 methods. In the first method, APP was considered a 2-class categorical variable, either anterior or posterior, using the subgroups described above. The percentage of improvement of NDI, VAS-N, and VAS-R was tested using univariable generalized linear regressions. The second method considered APP a continuous variable, and Pearson correlation was computed for each functional outcome. The Pearson correlation coefficient r varies between −1 (total inverse correlation) and 1 (total same sense correlation); 0 meant no correlation. The primary alpha risk was 0.05.
Analyses were performed using R software (R 3.6.0, R Foundation for Statistical Computing). This study was conducted per the ethical standards of the Declaration of Helsinki of 1964 and the Reference Methodology MR-004 of the French National Commission for Data Protection Commission (registration number 2225578).
Results
Data were compiled from 25 consecutive patients (14 women) with a mean age of 47 years (range: 28–73 years). The average follow-up was 25.9 months (range: 15 to 38 months). The FSU involved were 3 C4/C5, 13 C5/C6, and 9 C6.
The median anteroposterior depth of the vertebral endplates was 21 mm (mean 21.5 mm; Q1: 20 mm, Q3: 22.2 mm; range: 19 to 27 mm). The median crude offset from the vertebral center was 0.4 mm (mean: 0.3 mm; Q1: −1.5, Q3: 2 mm; range: −2.9 to 4 mm). The mean APP was 49%, ranging from 41% to 56% (Table 1, Figure 2). Ten patients had a posterior positioning, and 15 had an anterior positioning. The mean APP was 45.2% (95% CI, 43.2%–47.1%) in the anterior group and 54.1% (95% CI, 51.4%–55.3%) in the posterior group. The distributions of APP and crude offset distances are represented in Figure 2.
Characteristics of the cohort.
Distribution of the patients. The distribution was given for the anteroposterior positioning in percentage (top) and the crude offset distance from the center, in millimeters (bottom).
The mean preoperative VAS-N was 8.1 and turned to 2.7 postoperatively (P < 10−4). The mean preoperative VAS-R was 7.2 and turned to 2.5 postoperatively (P < 10−4). The mean preoperative NDI was 36.4% and turned to 13.9% postoperatively (P < 10−4). The mean ROM was 10.8° postoperatively.
There was no significant difference between the group anterior and the group posterior for all functional outcomes and the ROM (Table 2).
The difference in the functional outcomes, ROM, and sagittal alignment parameters according to the 2 groups.
The mean C2C7 Cobb angle was 11.6° preoperatively (range 1°–25°) and 15.6° postoperatively (range 0°–32°; P = 0.1). The mean SCA was 79° preoperatively and 74° postoperatively (P = 0.04). There was a significant correlation between SCA and C2C7 Cobb angle (r = −0.65, P < 10−4). There was no correlation between the APP and postoperative C2C7 Cobb angle (r = 0.16, P = 0.47) and postoperative SCA (r = −0.23, P = 0.31) and these 2 variables were not significantly different between the 2 groups.
The analyses were robust to the sensitivity analysis, and Table 2 presents the pooled results.
Eleven patients developed an HO by the LFU, 6 grade 1 and 4 grade 2. There was no significant association between HO and the functional outcomes, but the ROM was 10° less for grade 2 HO (P = 0.04). There was no association with the APP.
Discussion
Key Results
This follow-up of 25 single-level CDA using CP-ESP prostheses highlighted a significant improvement in functional outcomes over time. The prostheses tended to be anteriorly centered compared with the center of the upper plateau of the lower vertebra. However, there was no significant association between functional outcomes and positioning. A new sagittal alignment was reached postoperatively with a significantly different SCA, but there was no correlation between the sagittal alignment parameters and the positioning.
HOs occurred for 11/25 patients without impacting VAS and NDI.
Limitations
The main limitation is inherent to the retrospective studies. The power of this study is questionable with 25 enrolled patients. For instance, focusing on the NDI score, it would be necessary to enroll 230 patients to seek a significant difference with a power of 80%. However, this is very little difference—13.1 vs 14.6 at the LFU—likely without any clinical impact. There is probably no need for statistical tests to confirm such a slight difference.
Interpretation and Generalizability
The depth of the implant was not linked with any functional and radiological variable of our series. The permissive feature of the CP-ESP may explain this. There are limited data available in the literature for comparison. Yi et al found that too much depth was associated with sagittal radiological malalignment with a Bryan implant (unconstrained).29 Our series did not find the same conclusion. It would be interesting to assess the extent to which the height of the implant impacts the sagittal parameters; the CP-ESP is 1.5 to 3.5 times thinner than the Bryan,30 which could be an explanation for the different results. Particularly, too much height can stretch the posterior facet joints, which can be responsible for facet subluxation and pain.31–33 Wang et al34 did not find any correlation between sagittal alignment and NDI; our series reached the same conclusion.
The rate of HO in our series was lower than the findings of Marques et al, who found 84% of HO at 2 years, with CDA, and up to 92% at 5 years.35 The impact of APP remains unclear; Zeng et al found depth was a risk factor for HO.36 Our conclusion was different, with no correlation. In any case, the impact of HO on the functional scores seems minimal, and like Marques et al or Zeng et al,35,36 this series denied any correlation between NDI and HO.
This study assesses the static in the neutral position, which is a relevant parameter. Albert et al measured that neck is in a neutral position for approximately 70% of the time in a worker population.37 Le Huec et al showed that the static of the cervical spine has a high inter-individual variability,7 and the instantaneous center of rotation varies according to the FSU.4 This implies that there is a state of static alignment, and the unconstrained cervical prostheses have a particular neutral position,38,39 which Wachowski et al formalized as IHA0 (instantaneous helical axis).38 These last authors revealed an IHA0 significantly shifted dorsally with Prestige and Bryan implants, 2 unconstrained prostheses. The translation of the superior endplate of CP-ESP on neutral lateral radiographs was briefly evocated by Lazennec et al in a previous report.25 The prostheses in our series had a slight translation in the neutral position, meaning they have been involved in setting this neutral position. There is still limited information about the relationship between the neutral features of the implant, the alignment in the neutral position, and the functional outcomes. Our series suggests that the implantation of CP-ESP was followed by a new neutral state of sagittal alignment because the SCA was significantly different between the preoperative and postoperative periods, without any influence of the positioning.
Regarding kinematics, the ROM was not impacted by the positioning either, which is consistent with the findings of Kim et al.40 The beneficial role of the stiffness of a prosthesis is well identified when considering kinematics.11,20 However, the stiffness is also relevant in a static position to maintain the sagittal alignment in a neutral position, as illustrated by the slight translation of the superior endplate in our series. The core is challenged in a neutral position and during motion, so the resistance to wear is logically a critical parameter. The polycarbonate urethane core of the CP-ESP is highly resistant, even after sterilization.41 The comparison with the other unconstrained implants reveals less wear-related degradation for the CP-ESP compared with other brands.25,42 This means the CP-ESP is expected to have a good lifespan, even though it is challenging in static and dynamic positions, which remains to be confirmed clinically with further follow-up.
Conclusion
This series revealed that the functional scores and the ROM were independent of the APP of the CP-ESP. A new sagittal alignment was reached postoperatively with a new SCA; the sagittal alignment parameters were not correlated to the positioning. The development of HO occurred without a link to the positioning and impact on VAS and NDI. CP-ESP provided good postoperative functional outcomes, and its single-piece viscoelastic design was permissive enough to tolerate more significant variability in its implantation technique.
Footnotes
Funding The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests Marc Antoine Rousseau and Jean-Yves Lazennec are co-conceptor of the CP-ESP. No sources of financial and no material support were provided for this study.
Disclosures Baptiste Boukebous reports receiving compensation for attending meetings from Corin, Sanofi, and Coloplast. Mohamed Zoghlami reports receiving compensation for attending meetings from Sanofi. Cedric Maillot reports receiving compensation for attending meetings from Sanofi, Leo Pharma, and Smith & Nephew S.A.S. Jean Yves Lazennec reports receiving compensation for attending meetings from Sanofi and FH Orthopedics and is co-owner of the patent of CP-ESP. Marc-Antoine Rousseau reports receiving compensation for attending meetings from Corin, FH-Orthopedics, LEO Pharma, Sanofi, Digital Medical Hub, Johnson & Johnson Medical, Spine Innovations, and Pfizer and is co-owner of the patent of CP-ESP. The remaining authors have no disclosures.
- This manuscript is generously published free of charge by ISASS, the International Society for the Advancement of Spine Surgery. Copyright © 2023 ISASS. To see more or order reprints or permissions, see http://ijssurgery.com.