Historical Perspective
The implantation of the first motion-preserving artificial disc, the Charité, in the United States in 2000 sparked an enormous amount of enthusiasm among spine surgeons seeking an alternative to lumbar arthrodesis. In fact, the fervor for this new technology was so great that it precipitated the foundation of the Spine Arthroplasty Society (SAS) in the same year. Approval of this device by the US Food and Drug Administration in 2004 succeeded in also capturing the public’s attention. A 2005 New York Times article entitled “For chronic back pain, surgery may not be best answer” editorialized that “Non-fusion technologies such as artificial discs represent a paradigm shift in the surgical treatment of degenerative spine problems.”1 These were indeed heady times.
However, in the foreword to the enormous 791-page tome, Motion Preservation Surgery of the Spine, sponsored by SAS, Hansen A. Yuan (2007-2008 SAS president) penned these words of wisdom: “Although motion is not identical in degree at each level, it is also not the same in each of the 6 degrees of motion. Each anatomical region has a specific range and quality. Therefore, the preservation of motion is a daunting challenge for the scientists and surgeons.”2 These cautionary statements turned out to be prescient, as clinical adoption of disc arthroplasty—particularly in the lumbar spine—faced significant headwinds and was hampered by regulatory bodies and reimbursement entities.3
While artificial disc replacement seemed to pose an advantage in preserving natural motion and avoiding the adjacent level degeneration associated with fusion, there has been a decreasing trend in the use of disc arthroplasty in the lumbar region as compared with a rising trend within the cervical region.3 Although currently experiencing renewed popularity, the rate of lumbar disc replacement decreased by 85% from 2005 to 2017, while the rate of cervical disc replacement increased by approximately 800% during the same period according to the National Inpatient Sample database.4
Limitations of Lumbar Disc Arthroplasty
The primary complication of concern with lumbar disc arthroplasty is the development of facet joint degeneration, occurring in up to 50% of failure cases.5,6 The increased risk of facet joint degeneration with disc arthroplasty is associated with sagittal imbalance caused by improper placement of the implant and removal of the anterior longitudinal ligament. Revision burden for lumbar artificial disc replacement rose by 400% from 2005 to 2013, owing to an initial enthusiasm for the relatively new procedure but also a significant spike in revisions in 2012.7 Additionally, the presence of various contraindications and specific patient eligibility criteria has limited candidacy for lumbar artificial disc replacement to as low as 5% of a surgeon’s practice.8
As well intentioned as it was, in many ways, the introduction of lumbar disc arthroplasty into the surgical armamentarium was ahead of its time. In fact, it was only in 2024 that the National Center for Health Statistics issued specific International Classification of Diseases, 10th Revision, Clinical Modification diagnostic codes for lumbosacral discogenic pain associated with degenerative disc disease, an action spearheaded by the International Society for the Advancement of Spine Surgery.9 Additionally, in the ensuing 2 decades since the regulatory clearance of the Charité artificial disc, there has been a growing appreciation of the shortcomings and pitfalls of lumbar fusion based on empirical evidence and real-world utilization.10 Even the most ardent supporter of arthrodesis as an effective surgical option believes that disruption of the natural kinetic chain by fusing multiple vertebrae together is not a desirable objective. Also, we have developed a far better understanding of the biomechanical influence of isolated anterior column disc replacement on the function and degeneration of the posterior facet joints, underscoring the anatomical and molecular interdependence of the entire motion segment.11–13
Importance of The 3-joint Complex and Introduction of Lumbar Total Joint Replacement
An acknowledgment of the fundamental structural nature of the 3-joint complex where the intervertebral disc and the 2 facet joints at a single spinal level work collectively as a solitary functional unit dates to the pioneering work of Kirkaldy-Willis and Yong-Hing.14 They postulated that degeneration or dysfunction in 1 of these joints can impact the others, leading to a cascade of degenerative changes across the entire complex, a concept referred to as the “Yong-Hing” model. Subsequent imaging studies of morphological changes across the 3-joint complex have borne out this hypothesis.15
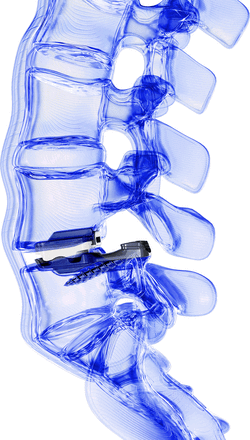
Inspired by the clinical success of total joint replacement (TJR) procedures of the large synovial joints of the appendicular skeleton and armed with a keen awareness of the need to address the entirety of the 3-joint complex of the lumbar spine functional unit, TJR of the lumbar spine was conceived.16 Like other TJR procedures in the hip and knee, for example, this revolutionary procedure couples a complete lumbar motion segment reconstruction with the implantation of a motion-preserving device using a bilateral transforaminal lumbar interbody approach to access the disc space.17 The implant, MOTUS (3Spine, Chattanooga, TN, USA), is the result of a 20-year research and development effort with the primary objective of replacing the motion segment with a fully functional device optimized to restore the primary kinematic characteristics of the 3-joint complex (Figure 1).
The MOTUS device (3Spine, Chattanooga, TN, USA).
TJR of the lumbar spine combines the clinical benefits of neural decompression with the maintenance of natural motion at the operative level with the implant functioning biomechanically as a new articulation for the resected disc and facets. Laminectomy, bilateral facet removal, and discectomy are used to achieve a wide central and bilateral decompression of the neural elements. The lateral annulus and anterior longitudinal ligament are preserved to maintain soft tissue tension, balance, and stability when disc height is restored. Additional surgical preparation includes a 3-column pedicle vertebral body osteotomy of the superior portion of the vertebral body and pedicle of the lower level to establish normal sagittal balance. The treated segment receives bilateral implants inserted along the axis of the pedicles, such that the midpoint of the implant is approximately 40% anterior to the posterior vertebral body which is consistent with the physiological center of rotation. Initial fixation is achieved by a keel and friction from a titanium plasma spray ingrowth surface at the implant/bone interface, as well as a retention screw in the caudal implant that passes obliquely through the pedicle and into the vertebral body of the caudal level. The flexion-extension motion of the disc and bilateral facet joints is reproduced by the device replacing all 3 joints of the lumbar motion segment (Figure 2).
Lateral view rendering depicting total joint replacement at the L4/L5 vertebral level.
The first human clinical experience with lumbar TJR was undertaken in 2007 with the implantation of 2 patients in South Africa.18 With 16 years of follow-up, there has been no resumption of symptoms, surgical revision, or evidence of degeneration or arthritic involvement at adjacent levels. Radiographic assessments, including computed tomography scan, magnetic resonance imaging, and plain radiographs taken in June 2023, displayed consistent and stable implant positioning, with no signs of implant wear, loosening, or failure. Both patients have been able to fully participate in all functions related to work, family, and recreation.
In a prospective clinical feasibility study, Sielatycki et al19 reported the 1-year clinical outcomes for a consecutive series of 52 TJR patients compared with a propensity-matched cohort of 156 transforaminal lumbar interbody fusion (TLIF) patients. Based on their analysis, the TJR group was significantly more likely to achieve clinically meaningful improvements in back function by Oswestry Disability Index, back pain, and leg pain than TLIF. For example, the patient acceptable symptom state defined as a 12-month Oswestry Disability Index value <20 and back and leg pain values <2 was achieved in 58% of TJR patients compared with 34% of TLIF patients (P = 0.003).
Conclusion: The Future of Motion Preservation
TJR of the lumbar spine is unique in that it synergizes the advantages of disc and facet arthroplasty using a single implant system, enhancing precision and personalizing treatment pathways. Utilizing the standard posterior operative approach with TJR allows for direct decompression of the neural elements, which may broaden the population of patients who can benefit from the procedure. The safety and effectiveness of TJR as an open surgical procedure are currently being investigated under an Investigational Device Exemption pivotal trial in the United States (ClinicalTrials.gov, NCT05438719). The procedural evolution of TJR to minimally invasive and endoscopic approaches is already under consideration. Lumbar TJR in many respects embodies the future of motion preservation. Coupled with the convergence of imaging, robotics, and artificial intelligence, TJR is positioned to continue the evolution of spine care, which is the core mission of the International Society for the Advancement of Spine Surgery. To determine the potential and accelerate the widespread clinical utilization of TJR, a concerted effort by surgeons, researchers, manufacturers, and regulatory bodies to generate and evaluate the evidence must be a singular goal.
Footnotes
Funding J.E.B. received financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests Jeffrey A. Goldstein reports a research grant from 3Spine, royalties/licenses from Globus Medical and Xtant, consulting fees from 3Spine and Globus Medical, support for attending meetings from 3Spine, and participation on an advisory board from 3Spine. Pierce D. Nunley reports a research grant from Orthofix, and participation on an advisory board from 3Spine. Ahilan Sivaganesan reports no conflicts. J. Alex Sielatycki reports consulting fees from Synergy Spinal Solutions. Anton Y. Jorgensen reports consulting fees from SI-Bone. Armen Khachatryan reports research grants from 3Spine, SpineArt, and Synergy Spine Solutions; consulting fees from Highridge Medical; honoraria for lectures from Highridge Medical; and a board position from Dymicron. Jeffrey A. Goldstein, J. Alex Sielatycki, S. Craig Humphreys, and Scott D. Hodges have stock in 3Spine. Additionally, Scott D. Hodges is an employee of 3Spine. Jon E. Block reports consulting fees from 3Spine and participation on an advisory board from 3Spine. Louis J. Nel reports no conflicts. Domagoj Coric reports royalties/licenses from Spine Wave, Acellus, Medtronic, and Globus; consulting fees from Spine Wave, Medtronic, and Globus; payment/honoraria from Spinal Elements; support for attending meetings/travel from Spinal Elements, Medtronic, and Globus; serving on the AANS Board of Delegates; and stock/stock options for Spine Wave, Premia Spine, and 3Spine.
- This manuscript is generously published free of charge by ISASS, the International Society for the Advancement of Spine Surgery. Copyright © 2025 ISASS. To see more or order reprints or permissions, see http://ijssurgery.com.